Technology
The Company’s proprietary tissue processing technology produces extremely strong, durable, and biocompatible tissue from xenogenic sources that demonstrate superior benefits compared to currently available biologic or synthetic products. Colorado Therapeutics products will be shipped Ready-For-Use / Off-The-Shelf in a dry state requiring no preparation or rehydration prior to use. Products in development include:
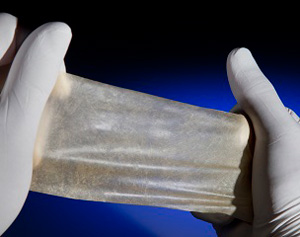 Soft-Tissue Repair / Abdominal Wall Reconstruction Applications: Colorado Therapeutics product utilizes cross-linked xenogenic pericardium which provides the strength and durability of synthetics with revascularization properties of non-cross-linked biologic products. The Company believes that the unique benefits of this product will be well suited for repairing hernia defects and in applications where current xenograft products are indicated. The Company’s proprietarily cross-linked pericardial product provides a dense connective tissue product that promotes remodeling, neovascularization (capillaries), and biological compatibility while resisting post-operative bulging with maintained strength and durability during the healing process.
Soft-Tissue Repair / Abdominal Wall Reconstruction Applications: Colorado Therapeutics product utilizes cross-linked xenogenic pericardium which provides the strength and durability of synthetics with revascularization properties of non-cross-linked biologic products. The Company believes that the unique benefits of this product will be well suited for repairing hernia defects and in applications where current xenograft products are indicated. The Company’s proprietarily cross-linked pericardial product provides a dense connective tissue product that promotes remodeling, neovascularization (capillaries), and biological compatibility while resisting post-operative bulging with maintained strength and durability during the healing process.
Pipeline
|
Colorado Therapeutics is exploring other applications utilizing the Company’s proprietary dry tissue processing technology including: Large Format Applications: for large abdominal wall reconstruction needs including for the tension-free applications/repair. Dura Repair Applications: for the use in dura mater repair to address cerebrospinal fluid (CSF) leaks. Dental Applications: for ridge augmentation and other periodontal and oral surgery applications. Orthopedic Applications: for applications in tendon/ligament augmentation.
|
Colorado Therapeutics’ devices are in development. No devices or other products are currently offered for sale by the company. Discussion of the technical features of products under development do not in any way constitute claims for clinical effectiveness or pledges of future performance.